
Author: Patrick John Oketch
Title: Water Audit Report Submitted to Uganda Breweries Ltd
Area:
Country: United States
Program:
Available for Download: Yes
View More Student Publications Click here
Sharing knowledge is a vital component in the growth and advancement of our society in a sustainable and responsible way. Through Open Access, AIU and other leading institutions through out the world are tearing down the barriers to access and use research literature. Our organization is interested in the dissemination of advances in scientific research fundamental to the proper operation of a modern society, in terms of community awareness, empowerment, health and wellness, sustainable development, economic advancement, and optimal functioning of health, education and other vital services. AIU’s mission and vision is consistent with the vision expressed in the Budapest Open Access Initiative and Berlin Declaration on Open Access to Knowledge in the Sciences and Humanities. Do you have something you would like to share, or just a question or comment for the author? If so we would be happy to hear from you, please use the contact form below.
For more information on the AIU's Open Access Initiative, click here.
INTRODUCTION
This assessment is aimed at evaluating the water systems at Uganda Breweries Limited, henceforth referred to as UBL, to validate and audit the current treatment regime, valuate area of concern to UBL, and map an improvement plan.
The following areas were looked into:
The Potable Water System
This treatment plant sources its water from lake Victoria. Its average flow is rated at 95M3/Hr.
Its principal composition is a mixing chamber sludge blanket clarifier and duplex sand filtration arranged in series.
The Chemical application is Aluminium Sulphate, Soda Ash and Calcium Hypo chlorite.
It was observed that;
The possible options of alleviating and addressing this cumulative problem is
To evaluate probable replacements, polyelectrolyte screening and jar tests have to be conducted in order to select the most suitable.
With jar test determining dosages its possible to submit a commercial proposal based on this.
Please note that only FDA approved polyelctrolyes will be screened.
2. The Water Pre-treatment System
This system is mainly used to precondition Boiler feed water system.
Water, ex Potable Water Treatment plant, is passed through a 1M3/Hr Sodium Zeolite softener at 10 bars. The water is then subjected into De-chlorination using Activated carbon filter. See Scheme 1 Below.
A residual chlorine test then determines the effectiveness of de-chlorination before the water is fed into the Feed Water Tank
Scheme 1
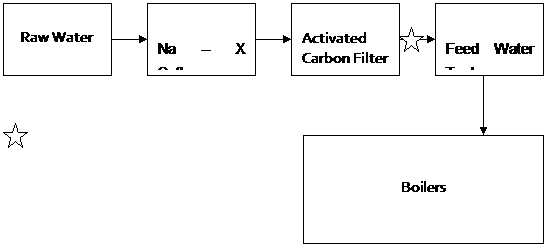
= Residual Chlorine Testing
This system was observed to be having a leaking priming valve at the feed water pump.
This was addressed immediately. Leaking valves can lead into air ingress into the feed water system. This contributes into high dissolved oxygen quantity that can lead into corrosion.
The feed Water tank was observed to be operating optimally in the following aspects
Feed water temperature is at 900 C
The De-aerators are venting off properly.
3. The Boilers
3.1 Fireside System
The principle fuel used is 125 cs HFO. Approximately 10M3 of fuel is combusted daily. The fuel is stored is bulk holding tanks. This is maintained at 400C.
Before combustion, the HFO is attemperation to between 800 – 1000C by direct injection of steam at 3.5 bars at the preheat chamber.
The Furnace combusts fuel at 2500 to 3000C.
The Air system is comprised of Induced Draft system pumping 100% environmental air.
It’s highly recommended to try and recycle 1/3 of hot stag gases into the combustion chamber.
3.2 Water Side
The general housekeeping is quite commendable.
The boilers, Ruby loose is rated at 14T/Hr and operates at 10 Bar while Thermax is a 13T/Hr boiler operating at 10 Bars as well.
Only one boiler operates at a time.
Condensate recovery was estimated to be above 80%
The boilers are effectively treated with Caustic Soda pH boosting, Anti-scallants, Oxygen scavenger and condensate control.
The basic observations and recommendations made here are to
RECOMMENDATIONS AND PROPOSED TREATMENT CHEMICALS
BOILERS
In the Boiler system, four main FDA approved products will be used:
Optisperse PO 5068.
The product is a blend of phosphate and unique scale inhibitor/dispersant blend. The dispersant is capable of reacting with the sludge/scale forming water constituents to enable them to be removed through the regular blow down thus keeping the heat transfer surfaces clean. The polymeric dispersant is particularly effective against iron.
Benefit summary
Highly effective scale inhibitor, dispersant and scale remover for Calcium, Magnesium, Iron, Copper etc – particularly effective against iron deposition.
Highly effective scale inhibitor, dispersant and scale remover for metal oxides.
Proven clean-up ability
Designed for high purity / high cycles system
Suitable for use at your operating pressures
Can provide in-service removal of existing scale, avoiding acid clean.
Chemically more stable and compatible with other treatments.
No interference with neutralizing amines.
Non-corrosive, safe to use even in case of overdosing or under dosing. Non-toxic, safe to handle and simple to dose with standard equipment.
Easy to test.
Optisperse PO 5068 keeps the boiler clean, maximizes heat transfer and ensures maximum efficiency thus reducing the operating costs.
Waterside deposit control in steam generating systems is the most effective means of ensuring optimum heat transfer. Control over waterside deposits is also the most effective means to help our customers keep their equipment on-line and reliable. One of the leading causes of unscheduled boiler shutdown is tube failure, which can be brought on by overheating in the high heat transfer areas when deposits are present. In today’s higher capacity, higher heat flux boilers, the potential for waterside deposit is greater than in the past.
Most elements dissolved or suspended in feed water have the potential of becoming a boiler deposit. Predominantly, waterside boiler deposits are comprised of two elements: Calcium and Iron.
Calcium
Resulting from either poorly operating pre-treatment equipment or from condensate contamination, calcium is the most prevalent deposit seen in the boiler systems. Due to the inverse solubility of calcium carbonate at high temperature and its insulating impact on boiler heat transfer, hardness levels in the feed water must be monitored and controlled. A 1/32 “ deposit thickness corresponds to a 2 % loss in operating efficiency.
Iron
Without a doubt, control over boiler water iron presents our industry with its greatest challenge. Iron may result from the poor feed water quality or from the corrosion in pre or post boiler systems and it is a potential boiler deposit-causing constituent. Iron scale of a given thickness will insulate a boiler tube 50 % more than calcium of comparable thickness. If Silica is bound in the deposit this insulating effect maybe 300 % that of Calcium.
Optisperse is able to provide very good dispersion on Calcium, Iron, Copper and Magnesium that exceeds the performance of most of the contemporary products.
Optisperse is incorporated into the structure of the alkaline scale as it is formed. In this way the normal regular crystals are completely distorted and there is little or no adhesion between the individual precipitated particles or between the particles and any solid (metal) surfaces in contact with them. The distorted form of the precipitates is further dispersed by the electrostatic dispersive action of the polymer, and the normal flow of water is sufficient to keep the small particles on the move until they are removed from the plant by normal blow-down. See the figures 1, 2, 3, 4 & 5.
Optisperse provides better iron and harness dispersancy compared to most other polymers. See Figures 6, 7 & 8.
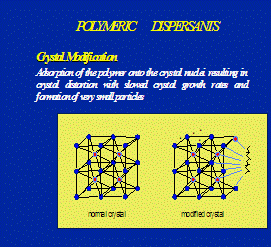
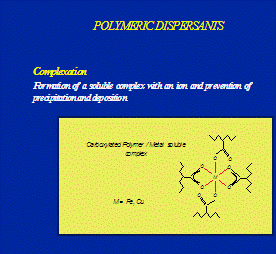
Fig. 1 Scale crystal complexation by Optisperse PO 5068 Fig.2 Scale crystal modification by Optisperse PO 5068

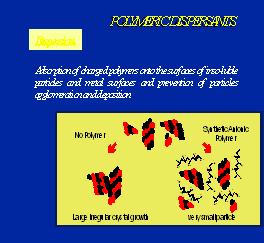
Fig. 3Scale crystal dispersion by Optisperse PO 5068Fig. 4 Scale crsytal structure before treatment
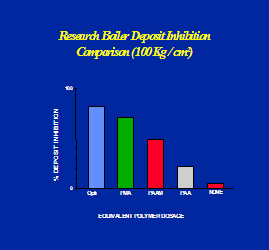
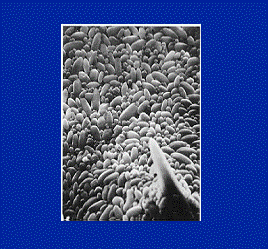
Fig 5 Scale crystal modification by Optisperse PO 5068 Fig 6 Boiler Deposit Inhibition Comparison
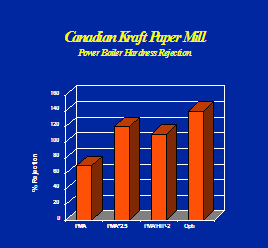
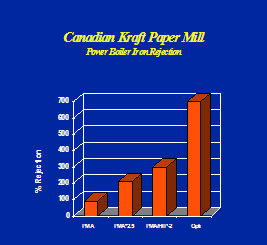
b). CORTROL IS 3000E: Oxygen Scavenger.
CORTROL IS 3000E is recommended for the rapid and complete removal of dissolved oxygen chemically from feed water and boiler water. The efficient removal of oxygen in the feed water ensures that no oxygen remains to contaminate the steam, thereby making the condensate less corrosive unlike the tannin based treatment
Benefits summary
Easy to use liquid.
Protects pre-boiler equipment.
Low toxicity.
Simple to control test.
Prevents Corrosion.
c). OPTISPERSE ADJ 5150: Alkalinity Builder & Organic Passivators
Dosing OPTISPERSE ADJ 5150 to the feed system for pH correction will help to promote a protective magnetite film, and hence to prevent corrosion.
OPTISPERSE ADJ 5150 will also produce the correct alkalinity levels within the boiler. This will assist in the prevention of calcium, magnesium and silicate deposits.
d). Steamate NA 0540E - Return Line Corrosion Inhibitor:
Steamate NA 0540E steam condensate treatment is a blend of volatile amines to provide positive corrosion protection by the direct neutralization of carbon dioxide. The reduction in corrosion products in the condensate is the result. Steamate NA 0540E is formulated to provide an ideal vapour - liquid distribution ratio, such that neutralizing amine is available to protect all parts of the steam/condensate system. Steamate NA 0540E is designed to balance and optimise individual characteristics of volatility and condensation, neutralising power, stability and solubility of its carbonate salt, to provide an effective product with minimum system losses for industrial and commercial boiler systems.
Advantages:
Prevents corrosion.
Reduces iron and copper pick up.
Convenient to dose.
Protects all parts of the condensate system.
FDA approved.
Treatment Programs
Boiler
Proposed Treatment Program
Product |
Daily Usage |
Monthly Usage |
6 Months Requirement |
Optisperse PO5068 |
3 |
90 |
540 |
Cortrol IS 3000E |
4 |
120 |
720 |
Optisperse ADJ 5150 |
1.5 |
45 |
270 |
Steamate NA 4220 |
2.5 |
75 |
450 |
*Monthly usage is taken that the boiler operates for 30 days in a month.
e) Fuel Oil System
System Details
Fuel Oil consumption/day – 10,000 litres.
Fuel Oil Quality - Class F Redwood
Technical Recommendations
We recommend the treatment of your fuel oil system with Fuelsolv OMG 2563 - a multifunctional approach offering the following features:
Excellent dispersancy
Combustion catalyst
Deposit Inhibition
Corrosion Inhibition
Environmental protection w.r.t NOx emissions.
The dosage of the product for your kind of fuel quality will be in the range of 200 to 500 ppm - typically about 400 ppm dosed in the fuel tank prior to oil delivery or mixed with the oil at delivery. It may also be metered into the oil burner line.
Product Capability
Below are defined some of the basic problems in a fuel oil system and brief explanations on how FuelSolv OMG 2563 is instrumental in addressing these concerns in fuel oil systems
Sedimentation
Sludge is composed of settled heavy agglomerates combined with suspended matter from an oil or liquid fuel. Sludge formation increases when fuel oils of different crudes are mixed. When fuel oils are heated in a tank to ensure good flow, the likelihood of sludge formation is increased. Sludge formation in the tank reduces the storage space for usable fuel and removes a part of the high energy containing components of the fuel. Frequent strainer cleaning is required to prevent high-pressure drops and ensure good flow. Sludge formation can also cause heater burner tip fouling.
Fuelsolv OMG 2563 is an excellent dispersant and is very effective in not only the prevention of sludge build-up but also removal of the existing sludge even at low dose rates thus resulting in:
Increased energy content in the fuel thus increasing the efficiency.
Maximum utilisation of the tank storage space
Clean feed lines, heaters and burners thus promoting good flow and good combustion.
Good flame patterns thus preventing flame impingement.
Operation at a minimum of excess air thus controlling slagging and high temperature corrosion.
Combustion
Combustion is normally a gaseous phase oxidation reaction, whether the fuel exists as a gas, liquid or solid. The notable exception to this is carbon, which can burn in a solid phase. For combustion to occur, the fuel is heated to drive off water, to cause it to decompose, and to vaporise its volatile components. Once the fuel is converted to a gas, it mixes more readily with oxygen from the combustion and allows the oxygen reaction to proceed.
The combustion rate of fixed carbon is much slower than the combustion rate of the volatile portion of the fuel, which burns in the gas phase. Therefore the cinders or ash present after combustion can contain a very high percentage of fixed carbon.
One way to make combustion more complete is to elevate the excess air; heating a greater volume of air to combustion temperatures lowers overall efficiency.
Another approach for pushing combustion further towards completion is the use of combustion catalysts. Combustion catalysts are primarily transition metals such as iron and Magnesium that catalyse the oxidation of carbon particles.
Particulate matter in the form of soot or coke is a by-product of the combustion process resulting from carryover of inert mineral matter in the fuel and from incomplete combustion. Primarily a concern on oil-fired units, particulate matter becomes apparent when fuel oil droplets undergo a form of fractional distillation during combustion, leaving relatively large carbonaceous particles known as cenospheres.
Cenospheres are porous, hollow particles of carbon that are virtually unaffected by further combustion in a conventional furnace environment. Cenospheres can also absorb sulfur oxides in the gaseous phase and there further contribute to the formation of acid smut.
Particulate matter from the combustion of fuel oil is a function of the fuel properties. Ash content of the fuel oil plays a significant role in forming such micron particulate emissions. These ultra fine particulate emissions are potentially more dangerous to the environment than larger particles, as they tend to stay suspended in the atmosphere. The fuel oil property most closely linked to forming cenospheres during the combustion process is asphaltene concentration. Asphaltenes are high molecular weight hydrocarbons that do not vaporise when heated. The combustion of fuel oils high in asphaltene content produces a greater quantity of large and intermediate size particulate emissions. Carbon residue, commonly determined by the Conraddson Carbon Test, is also a means of evaluating the tendency to form particulate matter during combustion.
Fuelsolv OMG 2563 contains a combustion catalyst which ensures the efficient burning of the fuel and minimising the formation of these large particulate emissions which can cause corrosion, fouling and environmental problems.
Problems caused by combustion
Carbon & Ashes
The formation of ashes and carbon causes an increase in the deposit accumulation on boiler surfaces, reducing exchange efficiency, increasing combustion chamber pressure and exit gas temperature. This causes a loss of efficiency due to higher exit gas temperature, load reduction, increasing excess oxygen, soot blowing frequency and shutdowns for boiler cleaning.
Vanadates
Most fuel oils contain varying contents of Vanadium. Vanadates form hard, low melting point deposits in the combustion chamber, superheater and air heater. These deposits being sticky in nature are not removed by soot blowing and are highly corrosive in nature and can affect on-line and idle boilers.
Sulfur Trioxide
Sulfur Trioxide gets formed in the system through oxidation of Sulfur and can cause corrosion in cold areas with skin temperatures below the acid dew point. It is instrumental in the blocking of the cold end areas due to liquid film formation of Sulphuric acid that incorporates dust particles, thus reducing exit gas areas. It leads to acute corrosion of the system below the dew point.
Environmental Problems
Combustion of the fuel leads to several environmental problems contributed by:
Dust load
Nitrous oxides
Sulfur Dioxide
Acid smutting - SO3
Carbon Monoxide
Corrosion Problems
All the above-mentioned factors also produce the following corrosion problems in the fuel oil system. These corrosion problems are related to the ash, Vanadium, Sodium and Sulfur content of the fuel oil, excess combustion air and combustion efficiency.
High Temperature Corrosion
The main cause of the high temperature corrosion is the formation of low melting point ash deposits. Sodium and Vanadium combine to form low melting point ash deposits, which cause corrosion. Sodium Vanadate complexes such as Sodium Vanadyl Vanadates (Na2O.V2O45V2O5) are the main concerns as they can have melting points as low as 540 deg C. Above melting temperature, deposits are sticky and extremely corrosive. Furthermore these deposits are very adherent, are very difficult to remove and can block superheaters if the tube spacing is small.
For corrosion, Sodium to Vanadate ratio (Na2O: V2O5) is important. When Na2O: V2O5=1:3 corrosion is catalysed. Preferential production of the 1,1,5 Sodium Vanadyl Vanadate could occur. Even with low Sodium and Vanadium content fuel oils, corrosion rate is rapid.
Corrosion mechanism proceeds with the oxidation of Vanadium to produce Vanadium Pentaoxide - V2O5 (melting point = 675 deg C). At metal temperatures above 450 deg c - 1,1,5 Sodium Vanadyl Vanadates are formed - Na2O.V2O45V2O5 (melting point = 625 deg C). Oxygen and Sulfur dioxide then further participate to yield corrosion of the metal.
With Oxygen
Na2O.V2O45V2O5 + Oxygen = Na2O.6V2O5
Na2O.6V2O5 + Me = Na2O.V2O45V2O5 + MeO
With Sulfur Dioxide
SO2 + Oxygen = SO3
SO3 + Na2O.V2O45V2O5 = Na2O.6V2O5 + SO2
Solutions to the High Temperature Corrosion problems
Reduce excess air to inhibit V2O5 formation and to aid V2O3 or V2O4 formation (melting point - 1950 deg C). This is often very difficult as it leads to incomplete combustion and thus intense sooting problems.
Use high Chromium content steel. This is a very expensive solution and is often not applied.
Use an appropriate additive (Magnesium additive) to produce high melting point ortho Vanadates (3 MgO.V2O5 - melting point = 1180 deg C). Deposits thus formed are dry and friable and are easily removed by soot blowing. The additive also inhibits the catalytic formation of SO3 thus reducing the acidic ash formation.
The colour of the deposits is also modified (White effect). High melting point complexes formed leads to increase in the combustion chamber temperature therefore the boiler produces lesser soot. Convective heat flux increases; superheater temperatures are easily attained and also reduces excess air. Reduction in the excess air leads to a reduction in the SO3 and Nox formation.
Greater cleanliness and reduced corrosion is attained. The reduction of the unburnt matter and the formation of the dry, friable deposits help maintain clean convective areas and economisers, especially with effective soot blowing. The presence of MgO in the deposits enables a non-acidic hydrolysis, so during the boiler shutdown, the metallic surfaces are protected against the acid attacks which typically occurs.
FuelSolv OMG 2563 is an excellent additive to inhibit high temperature corrosion resulting from the above-defined factors.
Low Temperature Corrosion
Low temperature corrosion results from the accelerated corrosion of surfaces due to condensed Sulphuric acid from flue gas. Deposition of Sulphuric acid occurs by the mechanical deposition of minute droplets or condensation on to the cold end surfaces or both. In the former case the flue gas is cooled below the acid dew point and sulphuric acid is condensed as a fog. In the latter case flue gas is above the acid dew point and the metal temperature is below the acid dew point. The dew point of the sulphuric acid in the flue gas depends on several factors e.g. fuel sulfur content, water content in flue gas, excess air and amount of SO3 produced.
Each of these factors elevates the dew point which is normally in the range of 125 to 150 deg C. Corrosion is worst at 20 to 30 deg C below the dew point and leads to sticky deposits and exfoliated corrosion products that can block the air heaters.
Sulphuric acid results from the formation of Sulfur Trioxide in the flue gas, which can result from the following routes.
1. Homogenous oxidation of SO2 in the flue gas by molecular oxygen ( post flame region)
SO2 + 1/2 O2 = SO3
2. Homogeneous oxidation of SO2 by atomic oxygen (in the flame and post flame region)
SO2 + O = SO3
3. Heterogeneous catalytic oxidation of SO2 to SO3
Oxides such as Fe2O3 and V2O5 act as catalysts
2 Fe2O3 + 6 SO2 + 3O2 = 2 Fe2 (SO4) 3
Fe2 (SO4) 3 = Fe2O3 + 3SO3
V2O5 + SO2 = V2O4SO3
V2O4SO3 + 1/2 O2 = V2O5 + SO3
4. Low temperature oxidation on surfaces below the acid dew point.
Sulphur Trioxide once formed condenses below the dew point forming H2SO4 and attacks iron as follows:
H2SO4 + Fe = FeSO4 + H2
When FeSO4 is formed the sulphuric acid has to diffuse through this layer to attack the iron. Diffusion is slow and the corrosion mechanism is thus reduced by its reaction product. When the pH is more than 3.5 free acid is minimal and the corrosion rate is very significantly reduced.
Solutions to the low temperature corrosion
The following factors lead to a reduction in the low temperature corrosion in a fuel oil system.
Reduce excess combustion air to inhibit Sulfur Tri-oxide formation.
Use low sulfur content fuel oil. This is an expensive solution.
Operate at high flue gas temperatures. (Expensive solution)
Improve combustion
Use an appropriate fuel additive. Fuelsolv OMG 2563 contains a magnesium additive which reduces the sulphuric acid corrosion by binding up the Sulfur Tri-oxide to form Magnesium sulphates thus neutralising the ash deposits and reducing the corrosion rate.
COSTS/CHEMICAL REQUIREMENTS
BASIS OF CALCULATION
300 Days a year operation
Fuel oil consumption – 10,000 litres per day
Dosage – 400 ppm
Annual Consumption 1,200Kgs
System Description |
Product |
Pkg. (kg) |
Fuel Oil System |
FuelSolv OMG 2562 |
23 |
4. COOLING SYSTEM
We recommend the treatment of the system with the following chemicals:
Continuum AT 3241: Corrosion & Scale Inhibitor
Continuum AT 3241 is a blend of inhibitors designed to control corrosion, scale formation and fouling, in an alkaline organic treatment programme, within open evaporative cooling systems. Continuum AT 3241 provides all the benefits of continuum technology in a single product.
The problem with the Zinc type inhibitors have been two-fold:
Zinc Hydroxide is only precipitated locally.
The film is not firmly bonded to the metal surface.
The result has been that in such a treatment large areas of the metal have been left exposed thus causing corrosion.
After extensive research, using electrochemical measurement, we have combined Zinc salts with a Phosphonocarboxylic acid in a specific ratio. We have also incorporated a novel polymer. The result is a cathanodic inhibitor, which can be used in high pH systems to control both scale and corrosion.
Advantages
Promotes the formation of a persistent protective oxide film on steel surfaces.
Controls fouling caused by suspended solids and inorganic scales, such as calcium carbonate.
Contains a unique halogen resistant azole, HRA, to inhibit corrosion of copper metallurgies.
Convenient liquid treatment.
Dosage: 85 ppm for Continuum AT 3241 in recirculating water.
BIOLOGICAL CONTROL PROGRAMME:
The following biological control program is designed to keep the open recirculating system from becoming contaminated with unacceptable levels of bacteria/algae. Fungi. Alternative use of two broad-spectrum biocides is recommended to prevent biological resistance. In addition we have recommended one bio-dispersant to control biological deposits & slimes.
Spectrus NX 1104: Biocide
Spectrus NX 1104 is a highly effective microbicide and slimicide extremely effective in controlling microbial growths over a wide spectrum of bacteria, algae and fungi. The biodispersant in Spectrus NX 1104 penetrates the microbial slimes and thus maximises the bio-kill. Designed to control the microbial growths in Industrial cooling water systems, the combination of non-oxidising microbicide and biodispersants in Spectrus NX 1104 ensures effective control over the formation of bio-films in the system. The treatment approach is effective over a wide range of pH and is an excellent supplement to the proposed Corrosion and Scale inhibitor
Features:
Effective at low concentrations
Controls a wide spectrum of bacteria, algae and fungi.
Penetrates microbial slimes
Low environmental toxicity
Inhibits bio-films.
Effective against Legionella pneumophilia
Effective at high pH.
Dosage: 80 ppm as slug feed to the tower basin twice per week.
Spectrus NX 1422: Biocide
Spectrus NX 1422 is a broad-spectrum microbicide slimicide and biodispersant effective against wide spectrum of bacteria / algae and fungi. It is especially effective against nitrifying and sulphate reducing bacteria at the recommended dose concentrations.
Features:
Broad-spectrum activity against bacteria.
Penetrates microbial slimes
Effective against Sulphate Reducing and nitrifying bacteria.
Low environmental toxicity.
Effective against Legionella pneumophilia.
Dosage: 100 ppm as slug feed to the tower basin once per week.
Control Parameters:
Continuum AT 3241: 20 ppm
Cycles of concentration: 8 cycles
Bacterial count: 250 RLU max.
CHEMICAL REQUIREMENTS
Cooling Water Treatment to comprise of:
Corrosion/Scale inhibitors
Biocides
System Description |
Product |
Dosage |
Cooling Water System |
Continuum AT 3241 |
4 Kgs/Day |
Spectrus NX 1104 |
2.0/Wk |
|
Spectrus NX 1422 |
2.0/Wk |
Pasteurizer
This will involve alternating oxidizing and non – oxidizing biocides in the system to maintain the desired toxicity levels and alleviate any probability of the microbes developing a resistance strain.
The proposed program is a safe, easy to use source of bromine, used for microbiological control in open evaporative and once through cooling systems. The bromine present in this product is in the form of an inactive bromide anion, Br-. It has to be oxidized hypobromous acid, HOBr, in order to be biocidal. This is achieved by activating the bromide with chlorine or sodium hypo chlorite solution. Hypobromous acid is effective for controlling microbial populations in cooling water,
This will be alternated with a proprietary water based non-oxidizing biocide, which is used to control microbial populations in open evaporative cooling systems. This is particularly effective against algae.
Please refer to the attached product fact sheets for more details.
Product Name |
Mode Of Action |
Pack |
Daily use |
Spectrus OX 1201 |
Oxidizing |
25 Kg |
2Kg (60 Kg) |
65% Calcium Hypochlorite |
Oxidizing |
45Kg |
0.5Kg (15 Kg) |
Continuum AT 3241 |
Anti-scallant |
23Kg |
2Kg (60 Kg) |
Notes:
5.The Fire Hydrant System
This will be a “Once Off” application of a Nitrite based corrosion inhibitor for closed loops.
The system charge requirement is 1500ppm.
The product recommended is Corrshield NT 4293.
See Attached product fact sheet for details.